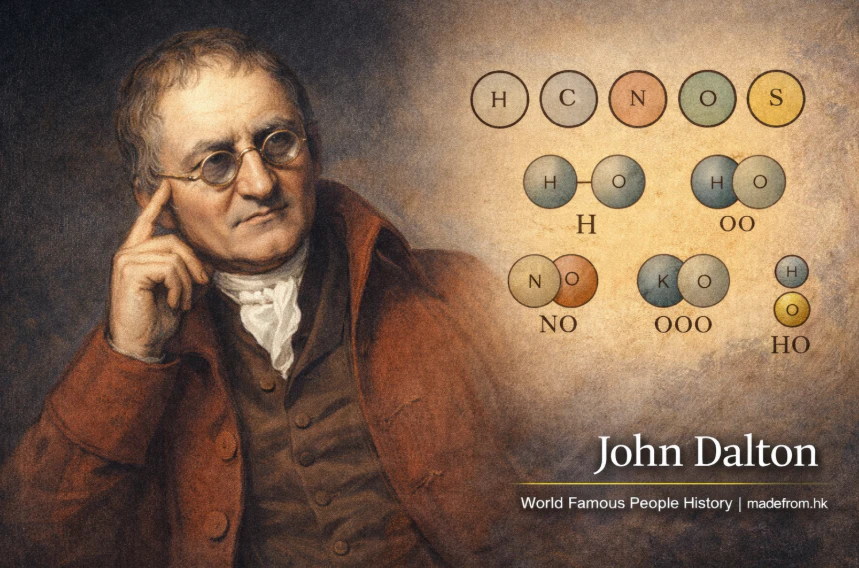約翰·道耳吞是英國著名科學家,提出現代原子理論,徹底改變人類對物質結構的理解,對化學與物理學發展影響深遠。
John Dalton was a pioneering English scientist whose atomic theory revolutionized the understanding of matter, laying the foundation for modern chemistry and influencing scientific thought for generations.
中文
John Dalton生於1766年英格蘭坎伯蘭郡,是一位在化學與物理學領域具有劃時代影響的科學家,被譽為「現代原子理論之父」。他出生於一個普通的貴格會家庭,自幼展現出對數學與自然科學的濃厚興趣,年紀輕輕便開始從事教學工作。道耳吞的科學研究跨越氣象學、物理學與化學,但最具影響力的成就是提出原子理論,徹底改變了人類對物質本質的認識。在19世紀初,道耳吞根據實驗與觀察提出:所有物質皆由不可分割的微小粒子——原子構成;同一元素的原子在質量與性質上相同,而不同元素的原子則具有不同特性;化學反應是原子重新排列組合的過程,而非創造或消滅物質。這些觀點雖然在今日已被進一步修正與發展,但其基本思想成為現代化學的基石。道耳吞亦提出「倍比定律」,指出當兩種元素形成多種化合物時,其質量比會呈現簡單整數比例,進一步支持原子存在的概念。他還建立了早期的原子符號系統,嘗試以圖形表示不同元素,雖然後來被更完善的化學符號取代,但在當時具有開創性意義。除了化學研究,道耳吞亦對氣體行為進行深入研究,提出氣體混合定律,即不同氣體在混合時各自獨立施加壓力,這一理論至今仍是物理與工程領域的重要基礎。此外,道耳吞還是最早研究色盲現象的科學家之一,他自身即患有色覺異常,並對此進行系統性研究,因此色盲曾被稱為「道耳吞症」。他的研究不僅體現科學探索精神,也展現個人經驗如何促進學術發展。道耳吞一生致力於科學研究與教育,長期在曼徹斯特從事學術活動,並成為當地科學界的重要人物。他的工作方式嚴謹而務實,強調實驗與觀察的重要性,對後世科學方法產生深遠影響。1830年代,他已被視為英國最具聲望的科學家之一,並獲得多項榮譽與學術地位。1844年,道耳吞在曼徹斯特逝世,享年78歲。儘管他的原子模型在現代科學中已被更精細的理論取代,但其核心思想仍是化學與物理學的基礎。他的貢獻不僅改變了科學界,也影響了工業革命時期對物質與能量的理解,成為人類探索自然世界的重要里程碑。
English Version
John Dalton was born in 1766 in Cumberland, England, and is widely recognized as one of the most influential scientists in the history of chemistry and physics. Raised in a modest Quaker family, Dalton displayed an early aptitude for mathematics and science, eventually becoming a teacher at a young age. His scientific pursuits spanned meteorology, physics, and chemistry, but his most enduring contribution was the development of modern atomic theory, which fundamentally transformed humanity’s understanding of matter. In the early 19th century, Dalton proposed that all matter is composed of tiny, indivisible particles called atoms; that atoms of the same element are identical in mass and properties, while atoms of different elements differ; and that chemical reactions involve the rearrangement of atoms rather than their creation or destruction. Although later scientific discoveries refined these ideas, Dalton’s framework laid the foundation for modern chemistry. He also formulated the law of multiple proportions, which states that when two elements combine to form different compounds, the ratios of their masses are simple whole numbers, providing strong evidence for atomic theory. Dalton introduced one of the earliest systems of chemical symbols, using circular diagrams to represent elements, marking an important step toward modern chemical notation. In addition to his work in chemistry, Dalton made significant contributions to the study of gases, proposing Dalton’s law of partial pressures, which explains how different gases in a mixture exert independent pressures. This principle remains essential in fields such as physics, engineering, and atmospheric science. Dalton also conducted pioneering research on color blindness, a condition he himself experienced; for many years, the condition was even referred to as “Daltonism” in his honor. His ability to turn personal observation into scientific inquiry exemplifies the spirit of empirical research. Throughout his life, Dalton was dedicated to education and scientific advancement, spending much of his career in Manchester, where he became a prominent figure in the scientific community. Known for his disciplined and methodical approach, he emphasized the importance of observation and experimentation, influencing future generations of scientists. By the 1830s, Dalton had gained widespread recognition and respect, receiving numerous honors for his contributions. He passed away in 1844 at the age of 78, leaving behind a legacy that continues to shape modern science. Although his atomic model has since been refined, the core principles he introduced remain fundamental to chemistry and physics. Dalton’s work not only advanced scientific knowledge but also supported the technological and industrial developments of his time, making him a key figure in humanity’s ongoing quest to understand the natural world.